 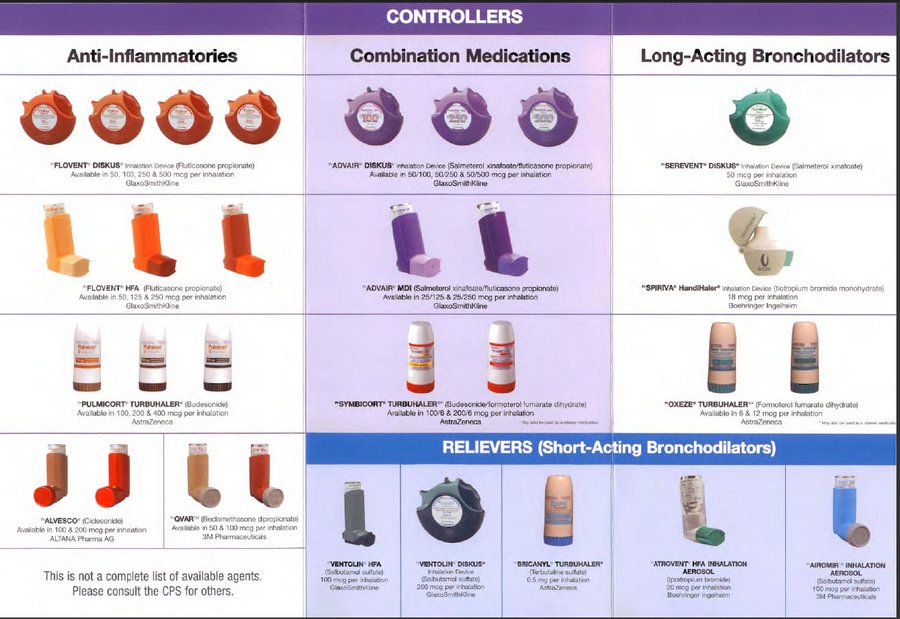
LABA and LAMA are types of bronchodilators. For people age 12 years old and older who are not well-controlled with an inhaled steroid, either a LABA or LAMA may be added. 
Long-acting bronchodilators can be either LABAs (long-acting beta2 agonists) or LAMAs (long-acting muscarinic antogonists). 
A blood sample should usually be taken 4–6 hours after an oral dose of a modified-release preparation. Long-acting bronchodilators are used regularly to open the airways and keep them open.Plasma-theophylline concentration should be measured 5 days after starting oral treatment and at least 3 days after any dose adjustment.Recommended blood monitoring on initiation of theophylline when required:.(brand name United States: Symbicort) 2 inhalations twice daily. Patients with good control (ACT>20 / CAT 225mg twice a day), or medications that can affect blood levels, or currently have active level monitoring, will require blood level monitoring if switched to theophylline. Usual doses of combination inhaled corticosteroids (ICS) and long-acting beta agonists (LABA) for COPD.NHS Lanarkshire Respiratory Team advice (February 2021): For information relating to interactions see BNF for details.ĭiscontinuation of aminophylline (Phyllocontin ®) tablets Unable to use multiple inhalers due to underlying conditions (e.g., cognitive deficits, dexterity or visual impairment, etc. Theophylline has a narrow margin between therapeutic and toxic effects therapy should be monitored. Documented difficulty adhering to 3-drug therapy via 2 separate inhalers (ICS/LABA + LAMA or LABA/LAMA + ICS) 2.Intravenous aminophylline is not a recommended drug in primary care having been superseded by nebulised beta 2–agonists.Prescribing by brand is recommended for oral formulations due to variations in bioavailability. However, if a LAMA inhaler by itself is not enough to control breathlessness or flare-ups, a combination inhaler with both a LAMA and LABA can be used.See below for advice on further management of patients currently prescribed Phyllocontin ® products.Please note - aminophylline preparations Phyllocontin ® Continus 225mg and Phyllocontin ® Forte Continus 350mg tablets have been discontinued ( MHRA Central Alerting System Feb 2021). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
